1 ▸).įace-centred cubic (neon, argon, krypton and xenon) and hexagonal close-packed (helium, beryllium and magnesium) crystal structures. Next, it might be considered surprising that solid helium, beryllium and magnesium all have the same crystal structure (Sluiter, 2007 ▸ Luo et al., 2012 ▸), which is hexagonal close-packed (h.c.p.) while neon, argon, krypton and xenon all have a face-centred cubic (f.c.c.) crystal structure (Sonnenblick et al., 1982 ▸ Moyano et al., 2007 ▸) (Fig. The question will then be, would solid helium above solid beryllium be regarded as equally irregular as the case of gaseous helium above solid beryllium? To be more specific, in the solid state, would we still support the idea of putting helium above neon because they are both noble gases? It is well known that solid helium has been obtained and characterized with its crystal structure being hexagonal close-packed (Donohue, 1959 ▸). However, for the sake of the observation in this paper, it is suggested that all the pure substances are considered in their solid state so that van der Waals forces become pronounced. In the majority of the common versions of the Periodic Table one can always find elements classified as solids, liquids and gases.
It can be seen that the choice of a Periodic Table of Pure Substances over the Periodic Table of Chemical Elements would probably cause an overcomplicated representation. But what about allotropes, the physical forms of chemical elements? We would need to either choose one of the allotropes or incorporate them all in one place, which does not seem rational. Which temperature and pressure do we choose for the representation? Which allotrope(s) do we prefer? As a matter of convention, we can choose standard temperature and pressure.

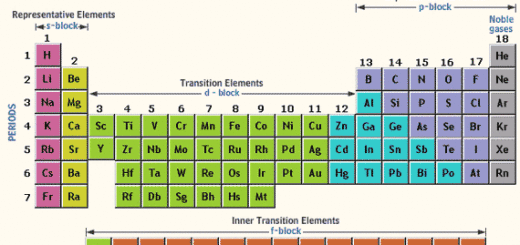
Numerous similar examples can be provided. Scerri fairly states, in case of the Periodic Table of Pure Substances, ‘one would probably not consider grouping together fluorine and chlorine, two green–yellow gases, along with a brown liquid bromine and a violet–black solid such as iodine’ (Scerri, 2005 ▸). Switching to the Periodic Table of Pure Substances would inevitably make us consider two further questions: (1) states of aggregation (2) allotropes. Thus, such a placement of helium transforms the Periodic Table of Chemical Elements into the Periodic Table of Pure Substances. According to the Gold Book, we then have one hundred and seventeen species of atoms and one pure chemical substance. Hence, the representation becomes inconsistent overall because the Periodic Table simultaneously adopts two different definitions of the chemical element. One hundred and seventeen of the known elements fit into those blocks however, there is only one element, helium, placed on top of the p-block as it is a noble gas. The IUPAC Periodic Table has four blocks of chemical elements: the s-, p-, d- and f-blocks, hence its whole body is based on electron configurations. Whichever representation of the periodic system is argued to be the optimal one (Leigh, 2009 ▸ Scerri, 2009 ▸), consistency of representation is the criterion that has to be met. The most common version of the revered icon of chemistry is the IUPAC Periodic Table of the Elements.

The one question that permanently accompanies the definition of the chemical element is the representation of the Periodic Table itself. Jensen suggested a definition (Jensen, 1998 ▸) which focuses on atomic nuclei rather on neutral atoms: (1) a class of nuclei, all of which have the same atomic number. Schwarz in his 2007 paper (Schwarz, 2007 ▸) argued that there are in fact three different definitions of a chemical element usually encountered: (1) a basic chemical element (2) a metallurgical element or simple material (3) an astrophysical spectroscopic element or elemental atom. A notable indication of the ongoing ambiguity is IUPAC’s Gold Book, which allows two different versions of the term ‘chemical element’: (1) a species of atoms – all atoms with the same number of protons in the atomic nucleus (2) a pure chemical substance composed of atoms with the same number of protons in the atomic nucleus. By 2019, one hundred and fifty years after Dmitry Mendeleev published the first successful version of the Periodic Table of the chemical elements, there was still no universal agreement regarding what a chemical element is.
